Precision oncology, the practice of leveraging genomic sequencing to guide cancer treatment, is now considered the standard of care. Yet for many cancer centers, the operational challenge is to turn critical insights from genomic sequencing into treatment decisions as quickly as possible.
The fragmentation of data compounds the difficulty of operationalizing insights at scale and creates complexity across tumor board workflows. Laboratory screening panel results, imaging results, and genetic sequencing results come from different entities. Treatment evidence resides in separate research knowledge bases. Tumor boards must manually reconcile information across multiple reports and systems, slowing treatment decisions, increasing variability in recommendations, and limiting the ability to scale precision medicine across the health system.
Here’s how evolv is helping comprehensive cancer centers transform fragmented precision oncology data into unified treatment intelligence that accelerates decisions, increases consistency, and enables measurement at scale.
A quick note before we dive in: A video embedded in this post provides a quick look at how evolv leverages Snowflake Cortex Code to spin up business outcomes quickly for our clients. We can deliver a true demonstration of tailored solutions just like this to solve your business problems. If you like what you see, reach out!
Why: The Precision Oncology Data Fragmentation Challenge
The Problem: Precision medicine generates rich molecular insights, but critical data exists in disconnected silos.
Clinical Context typically is contained in a patient’s Electronic Health Record (EHR) and includes data such as cancer diagnosis and tumor stage, patent demographics and medical history, imaging summaries and radiology reports, and lab results and pathology notes. Meanwhile, the Molecular Evidence, i.e., genomic sequencing results, gene variants and mutations, and clinical significance classifications, resides in a Laboratory Information Management System (LIMS) and/or external labs. Treatment Evidence is contained in research knowledge databases, including information such as therapy recommendations linked to specific mutations, clinical trial eligibility criteria, and evidence sources like OncoKB, CIViC, and published literature.
Since these datasets are not unified, tumor board members must manually reconcile information across multiple reports, switch between systems during case review, piece together patient context, molecular findings, and treatment evidence and then document decisions in separate systems with inconsistent formats.
This fragmentation creates measurable clinical and operational challenges, including delayed time to treatment and inconsistent recommendations, with different tumor boards interpreting the same data differently. It also often leads to limited clinician trust, since a lack of transparency in evidence sources undermines confidence. Fragmentation also means an inability to scale, since precision medicine cannot expand without standardization. There’s also no way to measure performance without unified data and visibility into metrics such as backlog of cases and time-to-decision.
As precision oncology becomes the standard of care, the operational challenge is clear: reduce data friction without disrupting clinicians or clinical workflows.
What: A Precision Oncology Treatment Intelligence Hub
Our Solution Approach: evolv designs Precision Oncology Treatment Intelligence Hubs that unify fragmented clinical, molecular, and evidence data into a single workflow that supports patient review, treatment recommendation, and decision documentation. We keep clinicians at the forefront of our design, and rather than replacing clinical systems or automating medical decisions, we create a governed data platform that supports tumor board review and treatment decision documentation.
Our approach centers on three integrated intelligence layers.
Layer 1: Patient Context Layer (Unified Patient Profile) We consolidate clinical and molecular context into a complete patient snapshot:
- Patient identifiers and cancer type/stage
- Biopsy confirmation and imaging summaries
- Genomic sequencing results with clinical significance
- Key mutations highlighted (actionable vs. non-actionable)
- Near real-time updates as new data arrives
Layer 2: Decision Support Layer (Treatment Recommendations) We connect molecular findings to evidence-backed therapy options:
- Recommended Therapies: Specific treatments matched to genomic variants
- Clear Rationale: Plain-language explanation of why each therapy is suggested
- Transparent Evidence: Direct links to knowledge bases (OncoKB, CIViC)
- Actionability Flags: Visual emphasis on mutations with targeted therapy options
- Traceability: Recommendations are directly traceable to source evidence, supporting transparency and trust during tumor board review
Layer 3: Governance & Audit Layer (Tumor Board Decisions) We capture what was decided, when, why, and by whom, thus creating an audit trail for each record:
- Decision status (approved, open, rejected)
- Decision notes documenting rationale
- Timestamps for compliance and quality tracking
- Historical timeline showing decision progression
- Consistent documentation format across all patients
How: Building Unified Precision Oncology Intelligence
The Implementation: evolv builds Precision Oncology Treatment Intelligence Hubs on Snowflake’s Data Cloud, creating a governed, scalable architecture that supports precision medicine at scale.
We ingest and structure data from multiple domains into a Unified Data Foundation, bringing together clinical data like patient demographics and biopsy findings with molecular data like genomic sequencing results and knowledge base integration, including gene-variant-therapy mappings with evidence levels.
Intelligence Layer 1: Unified Patient Profile
We create a comprehensive patient context view, a single patient snapshot that includes patient ID and demographics, the cancer type and stage, an imaging summary displayed in plain text, biopsy confirmation status, and key molecular findings with actionability flags. This profile includes near-real-time updates, offering an automatic refresh as new sequencing results arrive, dynamic highlighting of actionable mutations, and the potential for integration with existing EHR workflows. Additionally, it is all mobile-accessible, providing easy access for tumor board members.
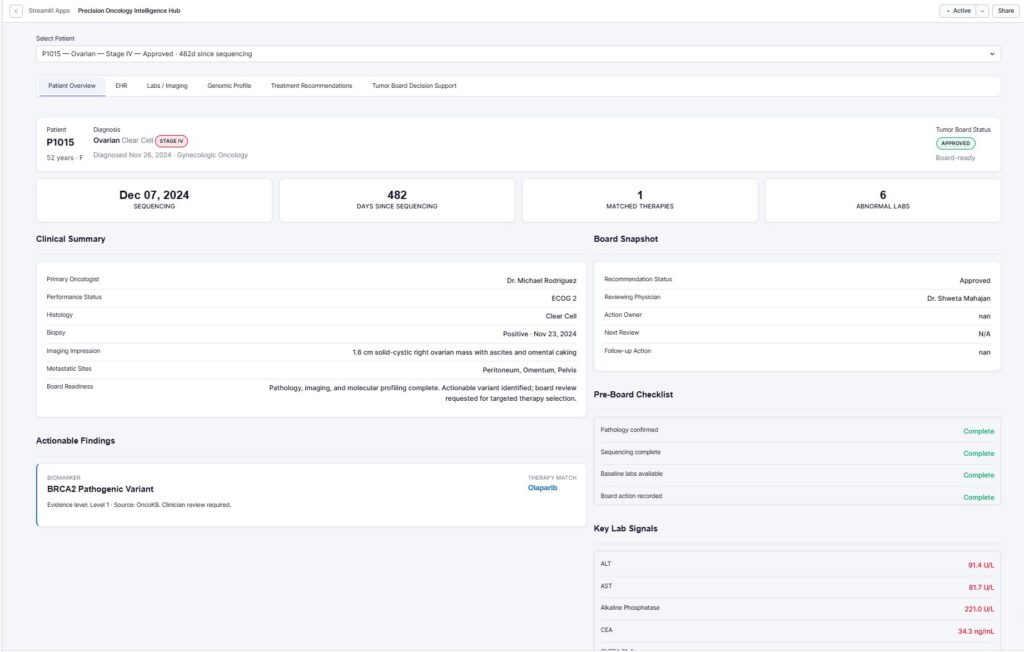
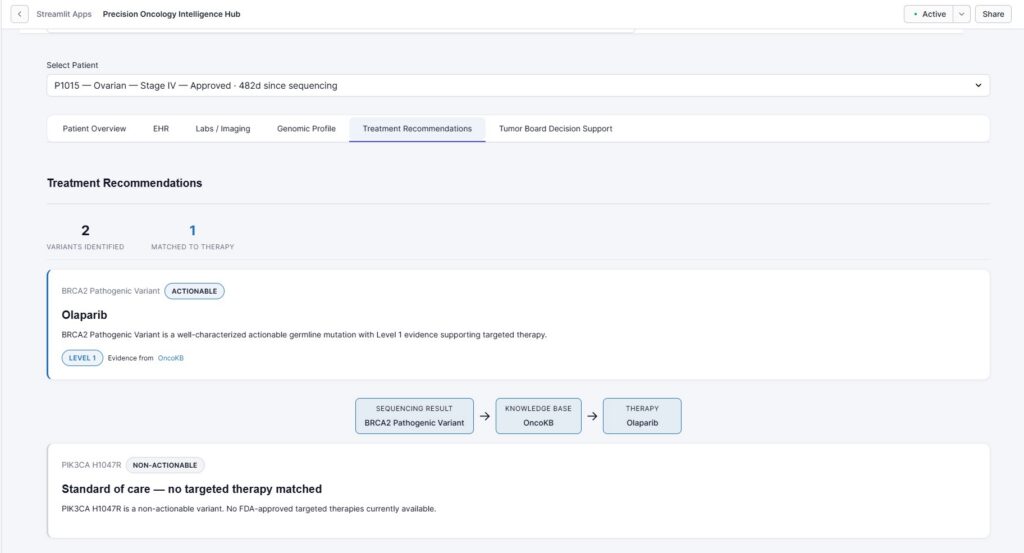
Intelligence Layer 2: Treatment Recommendations
We generate evidence-backed therapy recommendations, displayed in a Decision Support Table that includes recommended therapies, clear rationale (e.g., “EGFR L858R actionable mutation”) and evidence source with clickable reference. This builds transparency and trust, as every recommendation is linked to a trusted knowledge base, evidence levels are displayed (Level 1A, 2B, etc.), actionable mutations are visually emphasized, and non-actionable cases are clearly labeled.
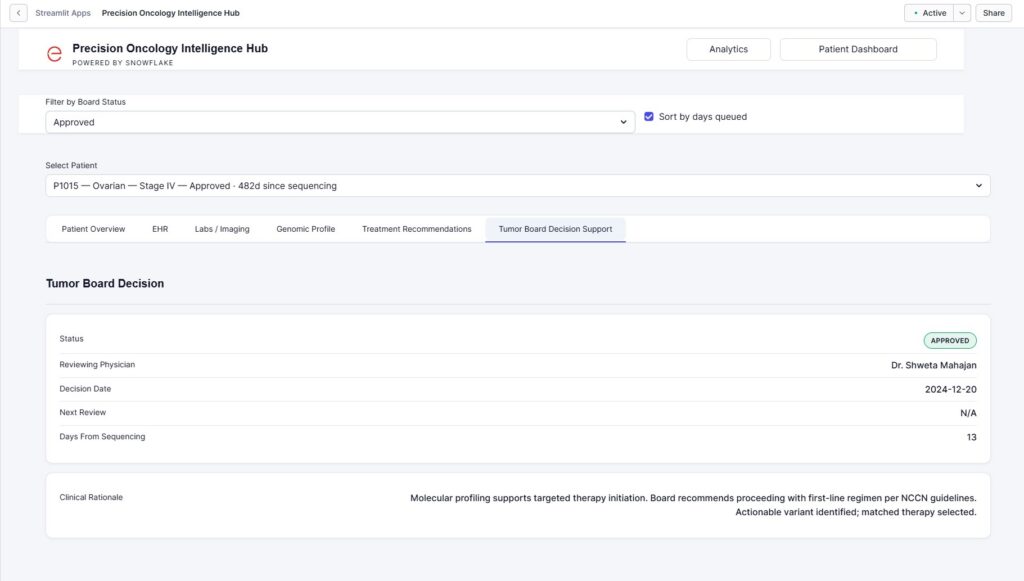
Intelligence Layer 3: Tumor Board Decision Tracking
We create an auditable record of treatment decisions, with decision documentation from the Tumor Board Decisions Panel. This includes decision status (think: approved, rejection, open), clinical rationale (free text documentation), decision date and timestamp, the reviewing clinician(s), and any required follow-up actions. We also include governance and an audit trail, complete from sequencing to therapy selection. Time-stamped decision records are also included, with consistent documentation format and a historical timeline showing decision progression.
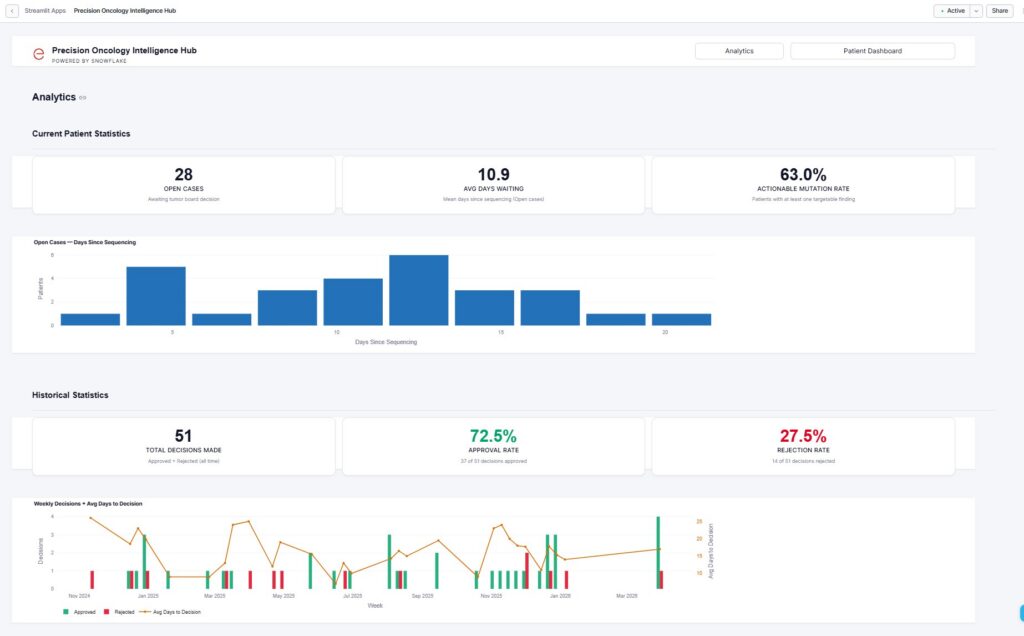
Integrated Intelligence Hub Dashboard
We compose all three layers into a single user experience, with a single-screen tumor board view that includes a patient overview (layer 1), therapy recommendations (layer 2), and decision history (layer 3). Executive Summary Metrics, such as the percent of patients with actionable mutations and the percent of recommendations approved are also included, alongside the average time from sequencing to decision.
Role-based views are also enabled, with oncologists viewing patient-specific molecular profiles, tumor board members given multi-patient views, research leads receiving aggregate genomic insights and outcomes, and IT leadership enabled to see data lineage and governance validation.
The Impact: Measurable Clinical and Operational Results
Precision Oncology Treatment Intelligence Hubs deliver quantifiable improvements across precision oncology operations:
✓ Faster Time to Treatment: A single, integrated dashboard replaces multiple system logins. Tumor boards are enabled to identify delayed cases and prioritize case reviews based on cancer staging and time since sequencing. Meanwhile, tumor board efficiency improves through unified patient context.
✓ Consistent, Evidence-Based Recommendations: A standardized knowledge base integration ensures consistency, transparent evidence sources increase clinician trust, and clear documentation of rationale supports clinical adoption.
✓ Scalable Precision Medicine Program: A standardized data model enables expansion across service lines, consistent processes support training and onboarding, and measurement infrastructure tracks program effectiveness.
✓ Auditable Decision Documentation: Providing complete records ensures quality assurance and compliance, time-stamped decision audit trails are included for regulatory requirements, and a consistent format simplifies chart review and analysis.
✓ Measurable Program Impact: Hubs allow for the tracking precision medicine utilization across the health system, measuring time from sequencing to therapy initiation, allowing for assessment of actionability rates and quantifying ROI of genomic testing investment.
The Bigger Picture
Implementing a unified treatment decision workflow is a fundamental transformation in precision oncology operations. By consolidating clinical context, molecular evidence, and treatment knowledge into a single governed platform, cancer centers can accelerate decisions, increase consistency, and scale precision medicine across the health system.
Precision Oncology Treatment Intelligence Hubs do not replace clinical expertise but serve as decision support systems. Oncologists can gain complete molecular context without switching systems. Tumor boards can make evidence-based decisions with transparent rationale. Researchers can track precision medicine impact at scale. Finally, and arguably most importantly, patients benefit from faster access to targeted therapies backed by the latest genomic science.
Ready to transform fragmented precision oncology data into unified treatment intelligence and decision workflows? Let’s talk about how evolv can help you build the governed data platform that accelerates decisions, increases consistency, and scales precision medicine across your health system. You can also check out our Precision Oncology Treatment Intelligence Hub service offering to learn more.
Shweta Mahajan is a Client Partner at evolv, with more than 17 years of consulting experience, primarily in healthcare. She is passionate about improving healthcare and partners closely with executive leaders to shape technology-enabled strategies, including data, analytics, and AI, to drive business outcomes and support patient care.
Shweta has led initiatives across clinical transformation, data modernization, and digital innovation, with a focus on aligning business priorities with practical execution. She brings cross-functional teams together to move initiatives forward and deliver results. Outside of work, she enjoys spending time with her dog and exploring her creativity through writing and the performing arts.




